Viability of Ischaemum rugosum Salisb. determined by the tetrazolium test
DOI:
https://doi.org/10.15517/am.v32i1.40318Keywords:
weeds, storage temperature, imbibition, seed cuttingAbstract
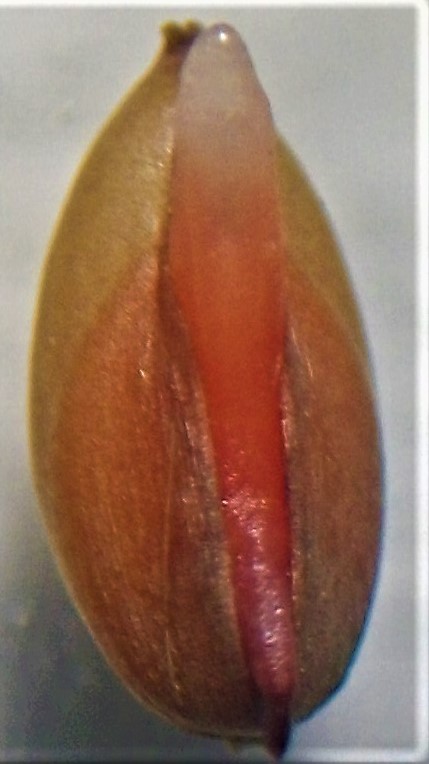
Introduction. The estimation of the viability of Ischaemum rugosum Salisb. with the tetrazolium test is a measure for the study of the weed’s biological dynamic. Objective. To evaluate the factors that affect the viability of I. rugosum Salisb using the tetrazolium test. Materials and methods. The work was carried out in the Laboratorio Oficial de Analisis de Calidad de Semillas del Centro para Investigaciones en Granos y Semillas of the Universidad de Costa Rica, San José, Costa Rica; in October 2015. I. rugosum Salisb. seeds were subjected to these treatments prior to the tetrazolium test: seed storage (room temperature: 23,9 °C and refrigerated temperature: 5 °C), imbibition (embedded and not embedded), caryopsid cut (with cut and without cut) and propagation unit (small caryopsis, large caryopsis, and spikelet). Seeds stained uniformly were counted as viable. Results. There was a significant interaction between storage temperature and propagation unit, the use of large caryopsis had a greater advantage over the small caryopsis (12.18 to 1) or the spikelet (181.27 to 1) and this result was stronger under room temperature. There was a significant interaction between propagation unit and cut, all the seeds required cutting so that tetrazolium could dye the tissues, the effect on the large seed was more conclusive than on the spikelet (1326.1 to 1) and that in the small one (7.39 to 1). The interaction of storage and imbibition temperatures and cutting and imbibition was significant. The seeds subjected to room temperature, embedded (41.26 to 1) and with cutting and imbibition (18.17 to 1) were more viable. Conclusion. The imbibition and cutting facilitated staining, the large seed and storage at room temperature influenced the viability of I. rugosum Salisb.
Downloads
References
Akanda, R. U., Mullahey, J. J., & Shilling, D. G. (1996). Environmental factors affecting germination of tropical soda apple (Solanum viarum). Weed Science, 44(3), 570–574. https://doi.org/10.1017/S0043174500094352
Alonso-Esquivel, M., Ortiz-López, Y., Ramos-Ramírez, R., Oliva-Diaz, H., & Capote-del Sol, M. (2011). Dormancia en semillas de papaya cv Maradol Roja durante el almacenamiento. Agronomía Mesoamericana, 22(2), 351–357. https://doi.org/10.15517/am.v22i2.11828
Aramendiz-Tatis, H., Cardona, C., Jarma, A., Robles, J., & Montalván, R. (2007). Efectos del almacenamiento en la calidad fisiológica de la semilla de berenjena (Solanum melongena L.). Agronomía Colombiana, 25(1), 104–112.
Awan, T. H., Chauhan, B. S., & Cruz, P. C. S. (2014). Physiological and morphological responses of Ischaemum rugosum Salisb. (Wrinkled Grass) to different nitrogen rates and rice seeding rates. PLoS One, 9(6), e98255. http://doi.org/10.1371/journal.pone.0098255
Benech-Arnold, R. L., Sánchez, R. A., Forcella, F., Kruk, B. C., & Ghersa, C. M. (2000). Environmental control of dormancy in weed seed banks in soil. Field crops research, 67(2), 105–122. https://doi.org/10.1016/S0378-4290(00)00087-3
Berjak, P & Pammenter, N. W. (2010). Manual de Semillas de Árboles Tropicales [Monografía]. Reforestation, Nurseries, & Genetics Resources. https://rngr.net/publications/manual-de-semillas-de-arboles-tropicales
Borza, J. K., Westerman, P. R., & Liebman, M. (2007). Comparing estimates of seed viability in three foxtail (Setaria) species using the imbibed seed crush test with and without additional tetrazolium testing. Weed Technology, 21(2), 518–522. https://doi.org/10.1614/WT-06-110
Doria, J. (2010). Generalidades sobre las semillas: su producción, conservación y almacenamiento. Cultivos Tropicales, 31(1), 74–75.
Giraldo-Cañas, D. (2010). Distribución e invasión de gramíneas C3 y C4 (Poaceae) en un gradiente altitudinal de los andes de Colombia. Caldasia, 32(1), 65–86.
Heap, I. (2020, July 1). The International Herbicide-Resistant Weed Database. WeedScience. http://www.weedscience.org/Home.aspx
International Rules for Seed Testing Association. (2014). Seed vigour testing. ISTA.
Hossain, M. A., Ishimine, Y., Akamine, H., Murayama, S., Uddin, S. M., & Kuniyoshi, K. (1999). Effect of burial depth on emergence of Panicum repens. Weed Science, 47(7), 651–656. https://doi.org/10.1017/S0043174500091281
Jarma-Orozco, A., Arvelaez, J., & Clavijo, J. (2007). Germinación de Ischaemum rugosum Salisb. en respuesta a estímulos ambientales y químicos. Temas Agrarios, 12(2), 31-41. https://doi.org/10.21897/rta.v12i2.1198
Jurado, E., & Westoby, M. (1992). Seedling growth in relation to seed size among species of arid Australia. Journal of Ecology, 80(3), 407–416. https://doi.org/10.2307/2260686
Leishman, M. R., Wright, I. J., Moles, A. T., & Westoby, M. (2000). The evolutionary ecology of seed size. En M. Fenner (Ed.), Seeds: the ecology of regeneration in plant communities (2a ed.) (pp. 31-57). Editorial. CAB International.
Marenco, R. A., & Santos, R. V. C. D. (1999). Wrinkledgrass and rice intra and interspecific competition. Revista Brasileira de Fisiologia Vegetal, 11(2), 107-111.
Ocampo-Sánchez, R. A. (1985). Incidencia de plantas indeseables en el cultivo de arroz (Oryza sativa) en el cantón de Aguirre y Parrita [Tesis de licenciatura no publicada]. Universidad de Costa Rica
Pabón, R. (1983). Algunos aspectos biológicos de la maleza falsa caminadora (Ischaemum rugosum) en los llanos orientales (Tesis de Maestría, Universidad Nacional de Colombia). Repositorio de la Corporación Colombiana de Investigación Agropecuaria. http://hdl.handle.net/20.500.12324/23911
Patil, V., & Dadlani, M. (2009). Handbook of seed testing. CABI Publishing.
Pérez, F. 2002. Material vegetal de reproducción: manejo conservación y tratamiento. Junta de Andalucía. http://www.juntadeandalucia.es/medioambiente/consolidado/publicacionesdigitales/80-402_MATERIAL_VEGETAL_DE_REPRODUCCION__MANEJO_CONSERVACION_Y_TRATAMIENTO/80-402/0_MATERIAL_VEGETAL_DE_REPRODUCCION.PDF
Rao, A. N., & Moody, K. (1995, July 24 to 28). Effect of seeding depth and soil moisture regime on emergence of Ischaemum rugosum and Echinochloa glabrescens. ResearchGate. https://www.researchgate.net/publication/286623927_Effect_of_seeding_depth_and_soil_moisture_regime_on_emergence_of_Ischaemum_rugosum_and_Echinochloa_glabrescens
Rao, N. K., Hanson, J., Dulloo, M. E., & Ghosh, K. (2007). Manual para el Manejo de Semillas en Bancos de Germoplasma (Manuales para Bancos de Germoplasma No. 8). Bioversity Intenational. https://www.bioversityinternational.org/fileadmin/_migrated/uploads/tx_news/Manual_para_el_manejo_de_semillas_en_bancos_de_germoplasma_1261_01.pdf
Rodríguez Quilón, I., Adam, G., & Durán, J. M. (2008). Ensayos de germinación y análisis de viabilidad y vigor en semillas. Agricultura: Revista Agropecuaria y Ganadera, 78(912), 836–842.
Vázquez-Yanes, C., & Cervantes, V. (1997). La reproducción de las plantas, semillas y meristemos. Biblioteca Virtual del Instituto Latinoamericano de la Comunicación Educativa. http://bibliotecadigital.ilce.edu.mx/sites/ciencia/volumen3/ciencia3/157/htm/lcpt157.htm
Westoby, M., Jurado, E., & Leishman, M. (1992). Comparative evolutionary ecology of seed size. Trends in Ecology & Evolution, 7(11), 368–372. https://doi.org/10.1016/0169-5347(92)90006-W
Downloads
Additional Files
Published
How to Cite
Issue
Section
License
1. Proposed policy for open access journals
Authors who publish in this journal accept the following conditions:
a. Authors retain the copyright and assign to the journal the right to the first publication, with the work registered under the attribution, non-commercial and no-derivative license from Creative Commons, which allows third parties to use what has been published as long as they mention the authorship of the work and upon first publication in this journal, the work may not be used for commercial purposes and the publications may not be used to remix, transform or create another work.
b. Authors may enter into additional independent contractual arrangements for the non-exclusive distribution of the version of the article published in this journal (e.g., including it in an institutional repository or publishing it in a book) provided that they clearly indicate that the work was first published in this journal.
c. Authors are permitted and encouraged to publish their work on the Internet (e.g. on institutional or personal pages) before and during the review and publication process, as it may lead to productive exchanges and faster and wider dissemination of published work (see The Effect of Open Access).